|
Ethnobotanical Leaflets 14: 648-53. 2010. Foliar Epidermal Morphology and Anti-Diabetic Property of Andrographis paniculata (Burm. f.) Wall ex. Nees
*T. R. Fasolaa , A. E. Ayodelea, A. A. Odetolab, and N. E. Umotoka
a Department of Botany and Microbiology, Faculty of Science, University of Ibadan, Ibadan, Nigeria b Department of Biochemistry, Faculty of Basic Medical Sciences, College of Medicine, University of Ibadan, Ibadan, Ibadan *E-mail:
Issued May 1, 2010
Abstract
The leaf epidermal features and anti diabetic property of Androgragphis paniculata were investigated. The epidermal cells are polygonal to irregular with curved to undulate anticlinal walls on both surfaces of the leaf. The leaf is hypostomatic with stomata restricted to the abaxial surface. Stomata type is axillocytic. The epidermal cells contain deposits of crystal oxalates while cystoliths are found in a few cells. The leaf extract was evaluated for hypoglycemic effects. There was significant hypoglycemic effect in alloxan induced diabetic rats following administration of the leaf aqueous extract. The result shows that the level of fasting blood sugar was dependent on the previous level of basal blood sugar in the four groups of rats used for this study. Treatment of the rats with A. paniculata showed a dose dependent relationship with the time it took for the blood of the rats to return to the basal blood sugar levels. The study supports the use of the plant in the management and treatment of diabetes. Key words: Andrographis paniculata; diabetes mellitus, antidiabetic properties, foliar epidermis. Introduction Andrographis paniculata (Burm.f.)Wall ex Nees is a small herbaceous plant of about 30-60m high. It is a member of the family Acanthaceae. A. paniculata is a native of India or Sri Lanka but now introduced to Nigeria and widely cultivated for its medicinal properties in south western Nigeria. The extract from the leaves and the roots is traditionally and widely used to treat various diseases such as cancer [Kumar et al., 2004; Pajagpal et al., 2003], upper respiratory tract infections [Poolsup et al., 2004; Coon and Ernst, 2004], filiariasis [Dutta and Sukul, 1982] and snakebites [Martz, 1992]. A. paniculata possesses hypoglycemic properties. An aqueous extract of A. paniculata has been shown to have hepatoprotective effects against carbon tetrachloride induced liver damage [Rana and Avadhoot, 1991]. The hypotensive activity of an aqueous extract of the plant has been demonstrated in rats [Zhang and Tan, 1996]. The plant is known to be beneficial following extracorporeal shock lithotripsy [Muangman et al., 1995]. The leaf extract of A. paniculata possesses antiplatelet aggregation [Amroyan et al., 1999], antifertility effect [Zoha et al., 1989] and uterus relaxation [Burgos et al., 2001].The main bioactive component of the plant is known to be a labdane diterpenoid called Andrographolide (Coon and Ernst, 2004). This study was undertaken to obtain information on the epidermal features of the leaf and to assess the hypoglycemic effect of A. paniculata leaf extract based on ancient reports of this property. Materials and Methods Plant material Samples of A. paniculata were collected from Kabba, Kabba Local Government Area in Kogi State of Nigeria and the Botanical Nursery of the Department of Botany and Microbiology, University of Ibadan, Ibadan, Nigeria. The plant materials were authenticated at the Herbarium of the Department of Botany and Microbiology, University of Ibadan, Nigeria (UIH) where voucher specimens were also deposited. Epidermal morphology Fresh leaves were obtained from the nursery of the Department of Botany and Microbiology, University of Ibadan, Ibadan, Nigeria while the dry leaves from the specimens from Kabba, were boiled in water for about five minutes to revive the cells. Methods of epidermal preparation follow Ayodele and Olowokudejo (1997; 2006). Descriptive terminology is according to Dilcher (1974). Preparation of the extract Leaves of Andrographis paniculata were collected early in the morning at about 9am. These were air dried at room temperature and later ground into powder form with the aid of an industrial blender. An aqueous extract was obtained by soaking 1g of the powdered leaves in 100ml of distilled water to produce 10mg/ml of aqueous extract. Experimental animals Thirty healthy Wistar rats (175-250g) maintained under normal or standard conditions were obtained from the animal house, Department of Veterinary Physiology, Faculty of Veterinary Medicine, University of Ibadan, Ibadan. Animal feeds were obtained from Caps Feed Mill Limited, Ibadan, Nigeria. Induction of diabetes mellitus in the rats with intramuscular alloxan. Ten percent alloxan dissolved in physiological saline was used to induce diabetes mellitus in the rats. Ten grammes of fresh sample of alloxan was weighed on a Mettlers balance and dissolved in 100ml of 0.15M physiological saline solutions to produce 10% alloxan. The dose administered was a standard dose of 100mg/kg weight of the rats. The exact volume of the drug administered was obtained using the general formula:
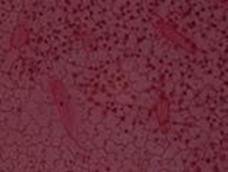
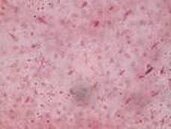
Volume to be administered = Weight of animal (g) x Dose of drug (mg/kg) 1000 x Concentration of drug (mg/ml)
The rats were divided into six treatment groups of five rats per group. The first four groups of rats were given intramuscular injection of 100mg/kg alloxan and Fasting Blood Sugar (FBS) was checked after 48 hours. The rats with fasting blood sugar between 100 500mg/dl were considered to be diabetic. Effect of plant extract on the diabetic rats The diabetic rats in each of the four groups were subsequently treated with different doses of (50mg/kg, 100mg/kg, 200mg/kg and 400mg/kg) of the plant extract twice daily for two weeks. This was administered orally with the use of oral canula. The fasting blood sugar was checked every two days by cutting a bit of the rats tails for a drop of blood and dropping the blood on a specified space on the glucometer and the reading was recorded. The fifth group (nondiabetic, not injected with alloxan had 400mg/kg of A. paniculata administered into them and the fasting blood sugar was recorded every two days for two weeks. The sixth group (control group) was fed with only feeds with clean water daily and their fasting blood sugar observed every two days and compared with the fasting blood sugar of the fifth group. Statistical analysis The results are expressed as MEANSEM. The students t test was used to determine the significance of difference between control and treated groups. The level of significance was taken as p < 0.05. Results and Discussion The results of this study are summarized in Figures 1- 3 and Tables 1-3. Figure 1 shows the habit A. paniculata as grown in the nursery of the Department of Botany and Microbiology, University of Ibadan, Ibadan, Nigeria, while figures 2 and 3 are photomicrographs of the adaxial and abaxial epidermis of the leaf of the plant. The epidermal cells are polygonal to irregular with curved to undulate anticlinal walls on both surfaces of the leaf (Figs. 2 and 3). The leaf is hypostomatic with stomata restricted to the abaxial surface. Stomata type is axillocytic (Fig.3) with one cell nearly enclosing both guard cells except for one free pole which is covered by two cells with a common anticilnal wall extending from the pole parallel to the long axis of the guard cells. The epidermal cells contain deposits of crystal oxalates while cystoliths are found in a few cells (Figs. 2 and 3). Table 1 shows the mean basal blood sugar levels of the four groups of rats and the mean fasting blood sugar levels after two days as a result of the induction of diabetes mellitus by intramuscular administration of alloxan. The result shows that the level of fasting blood sugar was dependent on the previous level of basal blood sugar in the four groups of rats. Treatment of the rats with A. paniculata showed a dose dependent relationship with the time it took for the blood of the rats to return to the basal blood sugar levels (Table 2). The maximum time was observed in Group 1 rats (i.e. those treated with 50mg/kg) which was more than 14 days. This was followed by Groups 2 and 3 (100 and 200mg/kg respectively). The shortest time of treatment was observed in the Group 4, those treated with the highest dose (400mg/kg). The administration of A. paniculata in normal rats showed that at the end of the two weeks, the mean fasting blood sugar had reduced from 51.4mg/dl to 42.0mg/dl (Table 3). In summary, the results of this study support the hypoglycemic properties of Andrographis paniculata as well as confirm the blood sugar reducing. References
Amroyan, E., Gabrielian, E., Panossian, A., Wikman, G. and Wagner, H. (1999). Inhibitory effect of andrographolide from Andrographis paniculata on PAF- induced platelet aggregation. Phytomedicine.: 6(1):27-31 Ayodele, A.E. and Olowokudejo, J.D. (1997). Systematic importance of leaf and epidermal characters in West African species of family Myrtaceae. Boletim da Sociedade Broteriana 68: 35-72. Ayodele, A. E. and Olowokudejo, J.D. (2006). The family Polygonaceae in West Africa : Taxonomic significance of leaf epidermal characters. South African Journal of Botany 72 : 442 - 459 Burgos, R.A. Aguila, M. J. Santiesteban, E.T., Sanchez, N.S. and Hancke, J. L. (2001). Andrographis paniculata (Ness) induces relaxation of uterus by blocking voltage operated calcium channels and inhibits Ca(+2) influx. Phytother-Res.: 15(3): 235-9 Coon, J. T. and Ernst, E. (2004). Andrographis paniculata in the treatment of upper respiratory tract infections: a systematic review of safety and efficacy. Planta Med. 70(4):293-8. Dilcher, D. L. (1974). Approaches to the identification of Angiosperm leaf remains. The Botanical Review 40:1-157 Dutta, A. and Sukul, N.C. (1982). Filaricidal properties of a wild herb, Andrographis paniculata. J. Helminthol 56: 81 - 84. Kumar, R.A, Sridevi, K., Kumar N.V., Nanduri, S. and Rajagopal, S. (2004). Anticancer and immunostimulatory compounds from Andrographis paniculata. J. Ethnopharmacol 92 (2-3):291-5. Martz, W. (1992). Plants with a reputation against snakebite. Toxicon. 30(10):1131-42. Muangman V, Viseshsindh V, Ratana-Olarn K and Buadilok (1995). The usage of Andrographis paniculata following extracorporeal shock wave lithotripsy ESWL). J Med Assoc Thai: 78(6): 310-313 Pajagopal, S., Kumar, R.A., Deevi, D.S., Satyanarayana, C. and Rajagopalan, R. (2003). Andrographolide, a potential cancer therapeutic agent isolated from Andrographis paniculata. J Exp Ther Oncol 3: 147-158. Poolsup, C., Suthisisang, C., Prathanturarug, S., Asawamekun, A. and Chanchareon, U. (2004). Andrographis paniculata in the symptomatic treatment of uncomplicated upper respiratory tract infection: systematic review of randomized controlled trials. J Clin Pharm Ther 29:37-45 Rana, A.C. and Avadhoot, Y. (1991), Hepatoprotective effects of Andrographis paniculata against carbon tetra-chloride induced liver damage. Arch Pharm Res:14 (1):93-95 Zhang, C.Y. and Tan, B.K. (1996), Hypotensive activity of aqueous extract of Andrographis paniculata in rats. Clin Exp Pharmacol Physiol. 23: 675-678 Zoha, M.S., Hussain, A.H. and Choudhury, S.A. (1989). Antifertility effect of Andrographis paniculata in mice. Bangladesh Med Res Counc Bull: 15(1):34-7.
Table 1. The mean values of Fasting Blood Sugar (mg/dl) of the four groups of rats after injection of alloxan.
Table 2. The effects of administration of different doses of different doses of A. paniculata in various groups of diabetic rats on mean values of Fasting Blood Sugar (mg/dl) taken on alternate days. Alternate days that Fasting Blood Sugar levels were taken
Table 3. The mean values of Fasting Blood Sugar (mg/dl) of Group 5 rats and Group 6 (Control group) rats taken on alternate days.
Alternate days that Fasting Blood Sugar levels were taken
Fig.1: Photograph showing the habit of Andrographis paniculata.
Fig. 2: Adaxial epidermal surface of A. paniculata.
Fig. 3: Abaxial epidermal surface of A. paniculata.
|