|
Ethnobotanical Leaflets 13: 301-309. 2009.
Studies on the Impact of Altitudinal Gradient on Ammonium Assimilatory Metabolism in Glycine max L. (Fabaceae) C. Rajasekaran1#*, T. Kalaivani2#, R. Jayakumararaj3, Abhijeet Singh4, V.R. Pusalkar5 and R. Marimuthu6
1Periyar University, Salem – 636 011, Tamil Nadu, IN 2Department of Biochemistry, A.V.S. College, Salem – 636015, Tamil Nadu, IN 3Department of Botany, Government Arts College, Dharmapuri – 636705, Tamil Nadu, IN 4M.N. Institute of Applied Sciences, Bikaner – 334 001, RAJ, IN 5Aruna Planta Medica, Salem – 636 004, Tamil Nadu, IN 6 Department of Botany, Government Arts College, Salem – 636 007, Tamil Nadu, IN *Corresponding author; C. Rajasekaran, #Present address: School of Biotechnology & Chemical Engineering, Vellore Institute of Technology University, Vellore -636 014, TN, India, Phone No: 9414903209; Email: [email protected]
Issued 15 February 2009 ABSTRACTSeedlings of Glycine max L. were grown, transplanted and acclimatized for 60 days at different altitudes (250, 400 and 1600 m). Response to shift in altitude was observed in the plants. Shoot length decreased with the increase in the altitude, while root length followed a reverse trend. Biomass accumulation in shoot and roots of G. max was the maximum at high altitude. Total soluble protein content was significantly high at low altitude in the shoot and the roots. Free tissue ammonia level in this species showed positive correlation with increasing altitude. Ammonium assimilatory enzymes viz., glutamine synthetase (GS), glutamate synthase (GOGAT) and glutamate dehydrogenase (GDH) were studied. GS/ GOGAT specific and total activity were altitude sensitive, whereas GDH activity exhibited inverse trend. Results indicate that there is a positive shift in ammonium assimilatory pathway in plants growing at high altitude. KEYWORDS: Ammonium Assimilatory Pathway; Glutamine Synthetase (GS); Gluatamate Synthase (GOGAT); Glutamate Dehydrogenase (GDH); Glycine max.INTRODUCTIONNitrogen metabolism in plants is a complex process, and is regulated by the form of nitrogen that is available to the plant (Magalhaes and Huber, 1989). Major source of nitrogen to the plant is ammonium, and is largely assimilated by the roots (Yoneyama and Kumazawa, 1974). Generally, plants prefer ammonium (NH4+) compared to nitrate (NO3-) and nitrite (NO2-) as ammonia is the starting point for nitrogen assimilation in higher plants. Internal source of ammonia is photorespiration and amino acid catabolism (Srivastava and Singh, 1987). However, high level of ammonia is toxic, therefore is converted to amino acids in plants (Miflin and Lea, 1980). Enzymes namely Glutamine synthetase (GS) (EC 6.3.1.2)/ Glutamate synthase (GOGAT) (EC 1.4.1.14) and Glutamate dehydrogenase (GDH) (EC 1.4.1.2-4) play a vital role in ammonia assimilation and detoxification. Off the three enzymes, GDH occupies a key role in plant metabolism. The ability of plant to acquaint to new environmental conditions depends upon its morphological adaptation and physiological response. In mountainous environment, variation in altitude offers wide variety of environmental conditions. In general, with increase in elevation, stressors such as temperature, pressure, light intensity, rainfall, partial pressure of metabolic gases are known to influence plant metabolism (Woodcock, 1976; Purohit, 1977). Like any other metabolic process, nitrogen metabolism is significantly influenced by variation in the altitude. Increased nitrogen content has been reported in plants with the increase in the altitude (Korner, 1989). This implies that that plants which are least influenced by altitudinal changes are much resistant to multiple stresses. Several workers have compared morpho-physiological response of plants to the change in the altitude (Bhadula et al., 1985; Rajasekaran et al., 1998; Rajasekaran, 2000). Ammonium assimilation play an important role in growth and development and has been extensively studied in various species. However, studies on ammonium assimilation in plants from the various climatic zones have been limited and no studies have been undertaken in Shervaroyan hills, part of Eastern Ghats, Tamil Nadu, India. The present work is an attempt to study growth behavior and analyze the activity of ammonium assimilation enzymes in Glycine max (L.) an annual leguminous food crop, grown at different altitudes in Shervaroyan hills. MATERIALS AND METHODSSeeds of Glycine max L. (Fabaceae) were sown in farmyard manure and garden soil in a ratio of 1:2 at Salem (250m) to raise seedlings. 40 day-old seedlings were transferred to experimental sites: Salem (250m), Kurumbapatti (400m) and Yercaud (1600m) in Shervaroyan hills of Salem, Tamil Nadu. Experimental plants acclimatized for 60 days at respective sites and growth performance was analyzed (shoot and root length and dry weight). For biochemical analysis, plant samples were frozen in liquid nitrogen after collection. Samples were homogenized in 0.1 M, Tris-HCl pH 7.5 containing 2 mM EDTA and 0.1% -mercaptoethanol, PMSF was added to prevent proteolysis. All the studies were conducted at 4°C. Total soluble protein was estimated using Bradford (1976) method. Free tissue ammonia level was estimated using method of Chaney and Marbach (1983). GS activity was determined by the method of Truax et al. (1994). One hundred μl of supernatant was incubated with 900 μl of assay buffer (0.1 M - imidazole-HCl, 65 mM - L- Glutamate, 4 mM - MnCl2, 0.75 ADP, 33 mM sodium arsenate and 17mM hydroxylamine, pH 6.8) at 30°C for one hour. The reaction was terminated by adding 1ml of stop solution (0.37 M FeCl3, 0.2 M TCA in 0.67 N HCl). After centrifugation, absorbance was determined at 540nm. Glutamylhydroxamate (Sigma Chemical Co, USA) was used to develop standard curve. A modified method of Dougall (1974) for the assay of NADH-GOGAT and NADH-GDH by following the rate of reduction of NADH at 340nm for 5 min. GOGAT assay mixture contain 20 mM Tricine, pH 7.5, 12.5 mM, α- ketoglutarate, 12.5 mM L-Glutamine and 0.15 mM NADH. Assay mixture for GDH contained 20 mM Tricine, pH 7.5, 1 mM α-ketoglutarate, 1 mM CaCl2, 100 mM NH4Cl and 0.15 mM NADH. All the estimations were carried out in triplicate (n = 3) following standard methods.
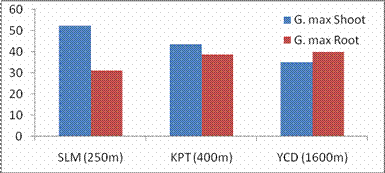
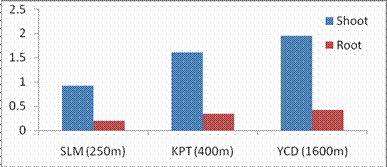
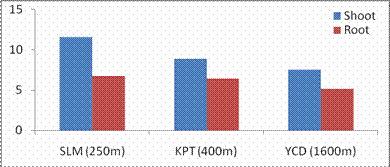
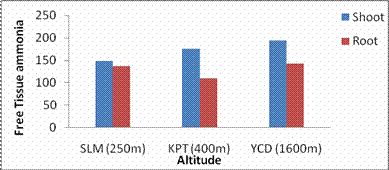
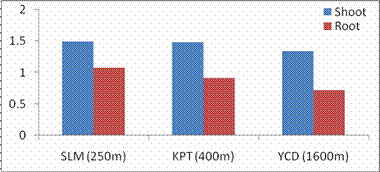
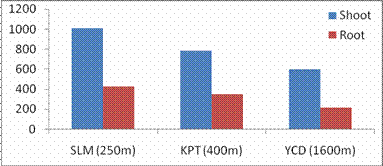
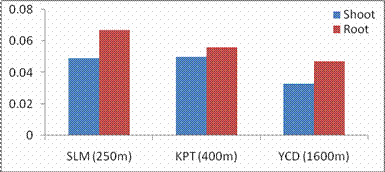
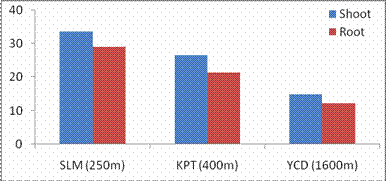
RESULTS AND DISCUSSIONPlant growth, biochemical and enzymes of ammonium assimilation in G. max were estimated. In G. max shoot and root length from three different altitudes is summarized in Fig. 1. Shoot and root length decreased with increase in altitude. Similar observation has been reported for different plant species by Nautiyal and Purohit (1980), Rajasekaran et al., (1998), Rajasekaran (2000). However, Bhatt and Purohit (1984) and Pankaj Prasad (1997) reported more root length than shoot at high altitudes due to non-availability of water in higher altitude and other factors such as soil, temperature. Sharma, (1980) reported dwarfism as an environment stress at high altitudes. Decrease in cumulative height and growth rate was steep in low land species than high land species (Todaria, 1980). Whereas shoot and root dry weight increased with the increase in the altitude (Fig 2). This could be due to higher concentration of carbon dioxide in the atmosphere (Purohit, 1998). Total soluble protein (TSP) content decreased with increasing altitude (Fig 3). However, free tissue ammonia (FTA) in G. max increased with increasing altitude (Fig 4). In roots maximum FTA was recorded at high altitude and minimum at low altitude. The present observations are in agreement with previous studies on Selinum vaginatum (Rajasekaran, 2000). Specific and total activities of GS are presented in Fig 5-6. GS specific and total activities of both the parts showed an inverse relation with the increasing altitude. Maximum activity was observed at the low and minimum at high altitudes. Variation in activity of GS under certain environmental conditions has been attributed to reassimilation of ammonia released during photorespiration (McNally et al., 1983; Wallsgrove et al., 1983). However, seasonal variation in GS of temperate deciduous tree leaves strongly indicated that decline in light intensity and temperature in late season accounted for drop in GS activity (Pearson and Ji, 1994), similar has already been reported in G. max and S. vaginatum (Rajasekaran et al., 1998; Rajasekaran, 2000). Decrease in GS activity under water stress in Albizzia stipulata and Oeugenia dalbergioides indicates that GS in both the plants is sensitive to water stress (Pankaj Prasad, 1997; Purohit, 1998), although in some plant species GS has been insensitive to water stress (Becana et al., 1984). FTA accumulation reflected in decreased GS activity in shoot and root (Figs. 4-6). Kamachi et al., (1992) reported that environmental conditions may induce FTA accumulation that may affect GS activity at high altitude. This is in accordance with previous reports (Cren and Hiral, 1999; Rajasekaran, 2000). This could be due to the fact that GS is less effective under high ammonia accumulation. Both, specific and total activity of NADH-GOGAT at three different altitudes is shown in Fig 7-8. NADH-GOGAT specific and total activity in shoot and root followed similar trend as in GS. NADH-GOGAT activity in shoots showed similar trend in low and middle altitudes. A decrease of 34% was observed at high altitude. Likewise, with the increase in the altitude decrease in shoot and root GOGAT specific and total activity was observed (Fig. 7-8). This indicates that FTA content in the tissues has complex regulatory effects on GOGAT activity as several stresses have been reported to be operative along different altitudes (Bhadula and Purohit, 1994; Pankaj Prasad, 1997; Purohit, 1998; Rajasekaran, 2000). Specific and total activities of GDH-amination in G. max (shoot and root) showed a positive correlation with increase in altitude (Fig. 9-10). Increased NADH-GDH activity has been reported along an altitudinal gradient (Pankaj Prasad, 1997; Rajasekaran et al., 1998; Rajasekaran, 2000). However, increased GDH activity is an indicator of detoxification of ammonia released during the breakdown of proteins and amino acids (Becana et al., 1984). In the present investigation, GDH-amination activity increased with the increase in the altitude (Fig. 9-10). Srivastava and Singh, (1987) reported that GDH pathway is active under certain nutritional and environmental conditions. Although, GS/GOGAT pathway is predominant in ammonia assimilation in higher plants, plants do switch over to GDH pathway under certain conditions of low energy and high ammonia (Yamaya, 1999; Rajasekaran et al., 1998; Rajasekaran, 2000). Specific activity of GS decreased at high altitude (Fig. 5), whereas at these altitudes GDH specific activity increased (Fig. 9). GDH plays a complementary role to GS/GOGAT cycle in synthesis of glutamate (Srivastava and Singh, 1987; Rajasekaran et al., 1998; Rajasekaran, 2000) or could catalyze the oxidation of glutamate to provide carbon-skeletons to the TCA cycle. At high altitude, FTA levels also were found to increase (Fig. 4). Increased levels of ammonia may have resulted in increase in de novo synthesis of GDH (Srivastava and Singh, 1987; Loulakakis and Angelakis 1990a; Watanebe et al., 1992). This may be one of the reasons for enhancement of GDH activities at high altitudes. However, investigations on isoforms, isoform patterns of ammonium assimilatiory enzymes, in-vitro studies using inhibitors of ammonium assimilatory enzymes along an altitudinal gradient is required to elucidate mechanism for this behavior.
ACKNOWLEDGEMENTSCouncil of Scientific and Industrial Research, Ministry of Human Resource Development, Government of India, New Delhi, is gratefully acknowledged for financial support (SRF- Extended fellowship, file no. 9/810(1)/2001-EMR- I) to CR. Authors are grateful to Dr. RSR Vice Chancellor, Periyar University, Prof. KVK for consistent encouragement to carryout this work. REFERENCESBecana, M., Aparicio-Tejo, P.M. and Sanchex-Diaz, M. (1984). Plant Physiol., 61: 653. Bhadula, S.K. and Purohit, A.N. (1994). Adaptational strategies of plants at high altitudes and future prospects for the conservation of biodiversity. Adv. Plant Sci. Res., 1:1-24. Bhadula, S.K., Pandey, O.P. and Purohit, A.N. (1985). Thermostbility and energy of activation of P-glycollate phosphatase and 3-PGA phosphatase in Selinum vaginatum Clarke grown at two altitudes. Photosynthetica, 19(3): 417-420. Bhatt, R.M. and Purohit, A.N. (1984). Morpho-physiological behaviour of two Anaphalis species from contrasting environments along and altitudinal gradient. Ind. J. Plan. Physiol., 27 (2): 130-137. Bradford, M.M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-257. Chaney, A.L. and Marbach, E.P. (1983). Modified reagents for determination of urea and ammonia. Chemical Biochem., 8: 130-132. Cren, M. and Hiral, B. (1999). Glutamine synthetase in higher plants: regulation of gene and protein expression from organ to cell. Plant Cell Physiol., 40: 1187-1193. Dougall, D.K. (1974). Evidence for the presence of glutamate synthase in extracts of carrot cell cultures. Biochem. Biophys. Res. Comm,. 58: 639-646. Kamachi, K., Yamaya, T., Hayakawa, T., Mae, T. and Ojima, K. (1992). Changes in cytosolic glutamine synthetase polypeptide and its mRNA in a leaf blade of rice plants during natural senescence. Plant Physiol., 98: 1323-1329. Korner, C.H. (1989). The nutritional status of plants from high altitudes, a worldwide comparison. Oecologia, 81: 379-391. Loulakakis, C.A. and Roubelakis-Angelakis, K.A. (1990a). Immuno-characterization of NADH-glutamate dehydrogenase from Vitis vinifera L. Plant Physiol., 94: 109-113. Magalhaes, J.R. and Huber, D.M. (1989). Ammonium assimilation in different plant species as affected by nitrogen form and pH control in solution culture. Fert. Res., 21: 1-6. McNally, S.F., Hirel, B., Gadal, P., Mann, A.F. and Stewart, G.R. (1983). Glutamine synthetase of higher plants. Evidence for a specific isoform content related to their possible physiological role and their compartmentation within the leaf. Plant Physiol., 72: 22-25. Miflin, B.J. and Lea, P.J. (1980). Ammonium assimilation. In: The Bio-chemistry of Plants. A comprehensive treatise: Amino acids and their derivatives (Eds. P.K. Stumpf and E.E.Conn). Vol. 5: 169-202. Academic Press. New York, London. Nautiyal, B.P. and Purohit, A.N. (1980). Morphophysiology of C3, C4 and CAM plants at different altitudes. Indian J. Plant Physiol., 23: 220-230. Pankaj Prasad. (1997). Eco-physiological aspect of fertilizer application and uptake in some nitrogen fixing tree species during early growth. D. Phil Thesis. H.N.B. Garhwal University. Srinagar (Garhwal). Pearson, J. and Ji, Y.M. (1994). Seasonal variation of leaf glutamine synthetase isoforms in temperate deciduous trees strongly suggests different functions for enzymes. Plant Cell and Environ., 17: 1331-1337. Purohit, A.N. (1977). Exploratory survey of floristic pattern in Garhwal Himalayas and a possible adaptability mechanism. The Himalaya, 1: 14. Purohit, I. (1998). Eco-physiology and biochemistry of nitrogen fixing tree species of Garhwal Himalaya. D. Phil Thesis. H.N.B. Garhwal University. Srinagar (Garhwal). Rajasekaran, C. (2000). Changes in the activities of enzymes of ammonium assimilation in plants along an altitudinal gradient. D. Phil Thesis. H.N.B. Garhwal University. Srinagar (Garhwal). Rajasekaran, C., Kuniyal, C.P., Prasad, P. and Bhadula, S.K. (1998). Growth and ammonium assimilation in Glycine max L. and Selinum vaginatum Clarke. Seedlings growth at three different altitudes of Garhwal Himalaya, India. Physiol. Mol. Biol. Plants, Sharma, M.C. (1980). Growth, development and productivity of Glycine soja, Dolichos lablab and Dolichos biflorus at different altitudes in Garhwal Himalayas. D. Phil. Thesis, H.N.B. Garhwal University. Srinagar (Garhwal). Srivastava, H.S. and Singh, R.P. (1987). Role and regulation of L-glutamate dehydrogenase activity in higher plants. Phytochem., 26(3): 597-610. Todaria, N.P. (1980). Growth behaviour and functional dynamics of plant species from different altitudes in Garhwal Himalaya. D. Phil. Thesis, H.N.B. Garhwal University, Srinagar (Garhwal). Truax, B., Lambert, F., Gagnon, D. and Chevrier, N. (1994). Nitrate reductase and glutamine synthetase activities in relation to growth and nitrogen assimilation in red oak and red ash seedlings; effects of N-forms, N concentration and light intensity. Trees, 9: 12-18. Wallsgrove, R.M., Keys, A.J., Lea, P.J. and Miflin, B.J. (1983). Photosynthesis, Photorespiration and nitrogen metabolism. Plant Cell Environ., 6: 301-309. Watanabe, M., Nakayama, H., Watanabe, Y. and Shimada, N. (1992). Induction of a specific isoenzyme of glutamate dehydrogenase during isolation and the first 48h of culture of Brassica napus leaf protoplasts. Physiol. Plant., 86: 231-235. Woodcock, R.G. (1976). Weather and Climate. M & E Hand Book. Eatover, Plagmouts. Yamaya, T. (1999). Molecular physiology of nitrogen utilization in plants. Jap. J. Soil Sci. Plant Nutr., 70: 255-258. Yoneyama, T. and Kumazaya, K. (1974). A kinetic study of the assimilation of 15N-labelled ammonium in rice seedling roots. Plant Cell Physiol., 15: 655-561.
Fig. 1. Changes in shoot and root growth patterns of G. max acclimatized at different altitudes.
Fig. 2. Changes in shoot and root dry weights of G. max acclimatized at different altitudes
Fig. 3. Changes in total soluble protein of shoot and root of G. max acclimatized at different altitudes.
Fig. 4. Changes in shoot and root free tissue ammonia levels of G. max acclimatized at different altitudes
Fig. 5. Changes in shoot and root GS (Specific) activity of G. max acclimatized at different altitudes.
Fig. 6. Changes in shoot and root GS (total) activity of G. max acclimatized at different altitudes.
Fig. 7. Changes in shoot and root GOGAT (Specific) activity of G. max acclimatized at different altitudes.
Fig. 8. Changes in shoot and root GOGAT (total) activity of G. max acclimatized at different altitudes.
Fig. 9. Changes in shoot and root GDH-amination (specific) activity of G. max acclimatized at different altitudes.
Fig. 10. Changes in shoot and root GDH-amination activity of G. max acclimatized at different altitudes.
|